Did you know that Truthout is a nonprofit and independently funded by readers like you? If you value what we do, please support our work with a donation.
This story was originally published by The 19th.
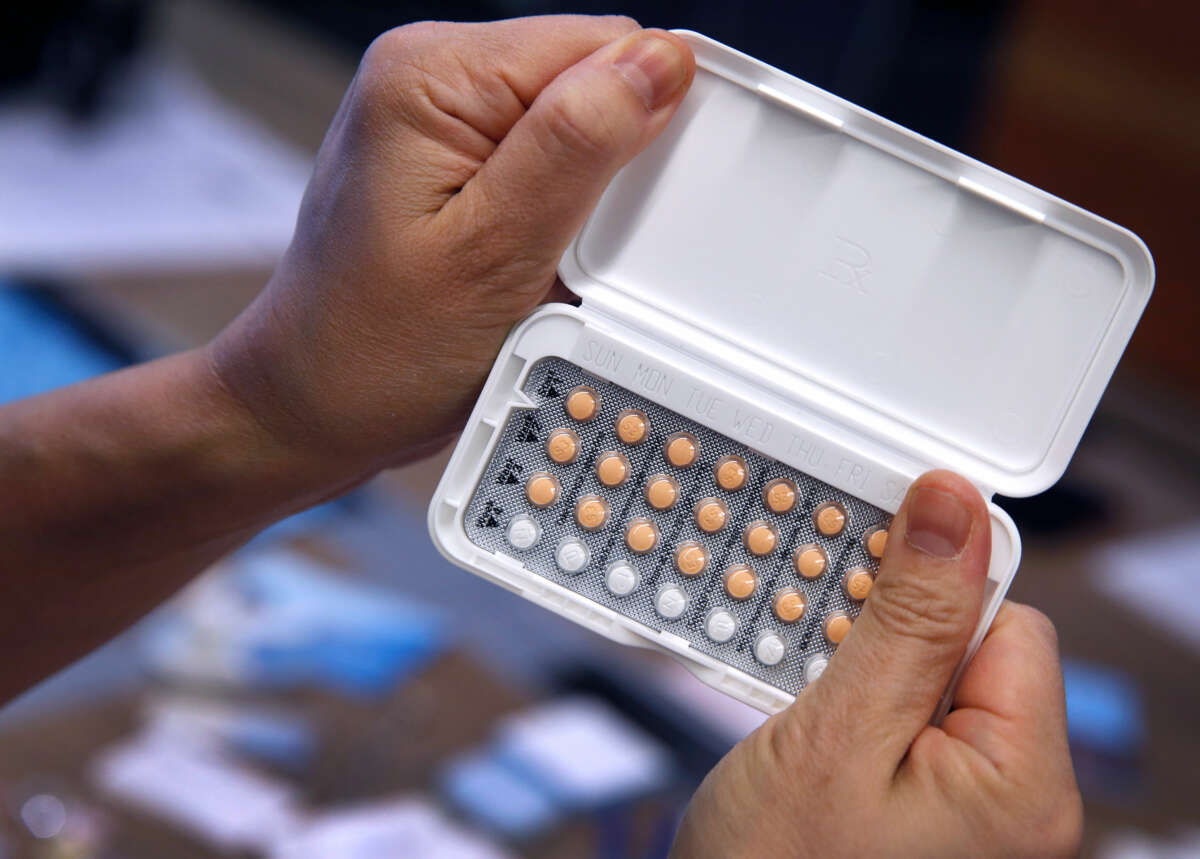
An influential advisory panel recommended that the Food and Drug Administration (FDA) approve an oral contraceptive pill for over-the-counter use without an age restriction.
While hormonal birth control is available without a prescription in many other countries, this medication, Opill, would be the first such option in the United States. The vote, conducted after a day-and-a-half discussion this week, is a significant step toward making birth control more widely available now that abortion is not federally protected.
The FDA is not obligated to accept the panel’s recommendation but is likely to do so, with a decision expected this summer. The vote means that hormonal birth control could be available without a prescription later this year.
In their review of the manufacturer’s data, FDA scientists surfaced concerns about whether people would know how to properly use the medication without the advice of a physician, highlighting in particular whether people would understand how often to take the medication, the proper way to consume it, the correct dosing, and what medical conditions might render them ineligible for the drug.
Opill, which is made of progestin, is not recommended for people who have had breast cancer. Some FDA scientists also worried that the drug may be less effective for people with higher body weights, who were not considered to the same extent when the drug was first approved in 1973.
But in their discussion this week, the group of scientists advising the FDA — including doctors and biostatisticians — argued that the value of making a safe and effective oral contraceptive more readily available outweighed those concerns. Making the pill an option without a prescription could make it easier for people who otherwise don’t have access to hormonal birth control: minors who are unable to to involve their parents, people who don’t have a regular health care provider, or those whose nearby clinics and health centers don’t provide oral contraceptives.
The over-the-counter birth control pill — which should be taken at the same time every day — is also more effective than other non-prescription methods, such as condoms or diaphragms.
“It’s really an access issue,” said Dr. Katalin Roth, a professor of medicine at George Washington University. “Making it over the counter is the right thing for women.”
Per data submitted by the drug’s manufacturer and analyzed by the FDA, most people included in the study did understand how to take Opill correctly. Some panelists noted that even people who receive oral contraceptives from a physician don’t receive robust counseling and don’t consistently take the pill at the same time every day.
Reproductive rights groups, as well as medical groups such as the American College for Obstetricians and Gynecologists and the American Medical Association, have endorsed approving the pill for over-the-counter use. Their concerns — that easy access to birth control is increasingly important now that states are legally allowed to ban abortion — were echoed in the panel discussion.
“The imperative for an oral contraceptive that’s available is now,” said Dr. Elise Berlan, an adolescent medicine specialist and associate professor at the Ohio State University College of Medicine.
Prescription birth control is supposed to be covered by health insurance, but plans aren’t required to cover over-the-counter methods. That means that Opill could still be out of reach for people with lower incomes, and especially Black women, Latinas and Native-American women, who on average earn the least on the dollar compared to White men. HRA Pharma, the drug manufacturer, has not indicated what it will charge for the drug. On average, a pack of hormonal contraceptive pills without insurance can cost between $20 and $50.
Orion Rummler contributed to this report.
An urgent fundraising appeal: 48 Hours to raise $20,000
Thank you for reading Truthout today. We have a brief message before you go —
Unfortunately, donations are down for Truthout at a time when media is under immense pressure. Trump is arresting journalists, Big Tech is censoring independent news, and economic conditions for media have been worsening for years.
Simultaneously, movement media is vital in the fight against Trump’s authoritarian reign. Our mandate to tell the truth, share strategies for resistance, and speak against fascism is ever more urgent in this deluge of political censorship. Yet, we are struggling to meet our publishing costs when our work is so urgently needed.
If you can support Truthout with a one-time or monthly donation, you will make a significant impact on our work. Please give today during our fundraiser (only 48 hours left).