Support justice-driven, accurate and transparent news — make a quick donation to Truthout today!
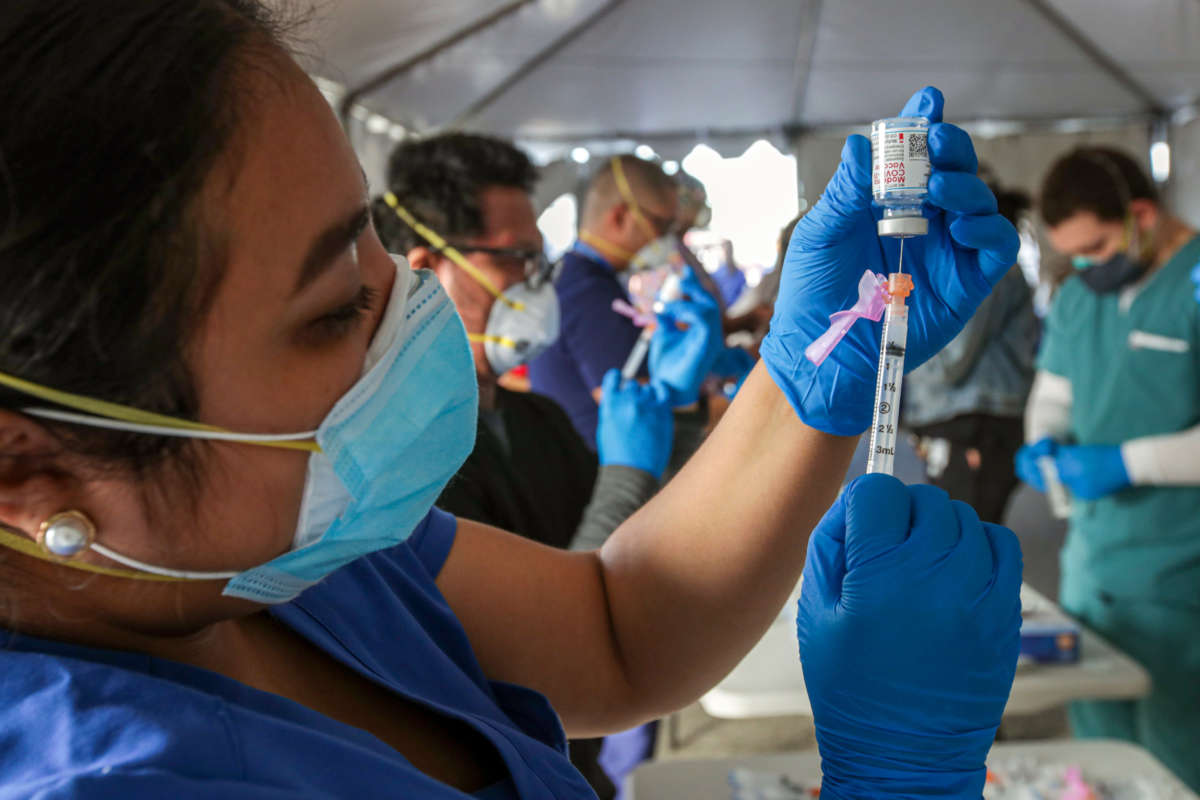
This week, the U.S. passed an encouraging milestone: More people in the U.S. have received their first dose of the coronavirus vaccine than have tested positive for COVID-19 since the virus first arrived stateside, according to Bloomberg. This news comes as a slate of other promising news on the pandemic has cropped up this first week of February.
The total number of vaccinated people surpassed the total number of people who had tested positive on Monday, according to Bloomberg’s count, at 26.5 million and 26.3 million, respectively. Since then, the gap has widened slightly, and there have now been 27.9 million vaccinations and 26.4 million cases in the U.S., according to the Centers for Disease Control and Prevention (CDC). The current vaccination rate in the U.S., Bloomberg says, is about 1.3 million doses administered a day, though some of the doses counted are the second dose.
With the numbers of vaccinated people growing, cases, hospitalizations and deaths have all dropped over the past week or so, according to The Atlantic. Case counts are still high, especially compared to lower rates from last year, but all three statistics are generally trending downwards from January’s highs.
Cases from last week are down at least 16 percent from the previous week, the COVID Tracking Project reports for The Atlantic. January had the most deaths yet from COVID, so there is still a long way to go, but this week’s drop in death counts is the first time deaths have dropped since October, they write.
Don’t miss a beat
Get the latest news and thought-provoking analysis from Truthout.
So far, the vaccine has mostly gone to medical workers and elderly people, which has led to promising downward trends in nursing homes: New health data is showing that cases in nursing homes, which have been among the deadliest places for the virus, dropped by 22 percent from December to January. A combination of prioritizing the vaccine for older people and holiday-related lockdowns in these facilities likely drove the trend downward.
And, on the vaccine front, more good news may be on the way. Since being inaugurated, President Joe Biden and his COVID team have been working to streamline vaccine distribution, announcing more vaccine purchases, increasing availability to states and unrolling test programs like sending vaccines directly to pharmacies beginning next week. Currently, only the Pfizer and Moderna vaccines have been available to the public, but more may be soon on the way.
On Thursday, Johnson & Johnson announced that it has officially submitted an application for emergency authorization to the Food and Drug Administration (FDA). Over the next weeks, the FDA will review the data with its vaccine advisory committee and vote on authorization on February 26, after which authorization would be announced shortly.
The company says that they are ready to ship doses as soon as it’s cleared. If authorized, this puts the Johnson & Johnson vaccine on track to start shipping at the beginning of March.
Johnson & Johnson announced the results of their vaccine trials last week. Though their vaccine isn’t as effective in preventing moderate to severe infection as those developed by Pfizer and Moderna, it still was 100 percent effective at preventing hospitalization and death in the trials. It’s easier to transport and is a one-shot regimen instead of Pfizer and Moderna’s two-dose regimen, which could free up time and money for more vaccinations.
On Friday, a new study showed the vaccine developed by AstraZeneca and the University of Oxford in the U.K. protected people against the new, more contagious variant of the virus that was first found in Britain. The AstraZeneca-Oxford vaccine was also found to prevent all hospitalization and death and is 84 percent effective against the original strain of the virus and about 75 percent effective against the strain found in the U.K. However, the study has yet to be peer reviewed.
Though the AstraZeneca-Oxford vaccine has been approved for use in Europe, AstraZeneca will have to finish clinical trials in the U.S. before it can apply for authorization from the FDA. The authorization will likely not take place for another month or two, and officials in December said that the U.S. will likely have to wait until April to use the vaccine.
An urgent fundraising appeal: 72 Hours to raise $25,000
Thank you for reading Truthout today. We have a brief message before you go —
Unfortunately, donations are down for Truthout at a time when media is under immense pressure. Trump is arresting journalists, Big Tech is censoring independent news, and economic conditions for media have been worsening for years.
Simultaneously, movement media is vital in the fight against Trump’s authoritarian reign. Our mandate to tell the truth, share strategies for resistance, and speak against fascism is ever more urgent in this deluge of political censorship. Yet, we are struggling to meet our publishing costs when our work is so urgently needed.
If you can support Truthout with a one-time or monthly donation, you will make a significant impact on our work. Please give today during our fundraiser (only 72 hours left).