 via Shutterstock )” width=”400″ height=”267″ />(Image: Pills and Money via Shutterstock)
via Shutterstock )” width=”400″ height=”267″ />(Image: Pills and Money via Shutterstock)
This story was co-published with The Boston Globe.
Pharmaceutical companies pay for the clinical trials that Dr. Yoav Golan conducts on antibiotics at Tufts Medical Center.
They also pay him tens of thousands of dollars a year to give speeches and advice on behalf of their drugs.
If Golan worked at some teaching hospitals, he would be barred or severely restricted from accepting both research funding and personal payments for promotional speaking or consulting from drug makers. These hospitals fear the money could influence clinical findings, or at least create the appearance of a conflict of interest.
Yet Tufts and many other academic medical centers allow doctors to accept overlapping payments — and some doctors still take them.
A ProPublica analysis shows that more than 1,300 practitioners nationwide received both research money and speaking or consulting fees from the same drug maker in 2012. All told, they received more than $90 million in research grants — plus nearly $13 million for speaking engagements and another $4 million for consulting.
Critics say doctors who conduct a clinical trial while accepting personal payments from the company sponsoring the study can feel beholden to the drug maker.
“The pharmaceutical company has a paramount stake in a favorable outcome. The [research] grant recipient has a stake in a favorable outcome and the honorarium recipient or consultant has yet another stake in the outcome,” said David Rothman, director of the Center for Medicine as a Profession at Columbia University. “It’s not only my lab. It’s my mortgage.”
ProPublica used its Dollars for Docs database, which tracks payments to practitioners by 15 drug companies, to conduct the review. Not every company discloses all types of payments — research, speaking and consulting — or distinguishes between the types. The analysis covered the nine companies that disclosed payments in this form.
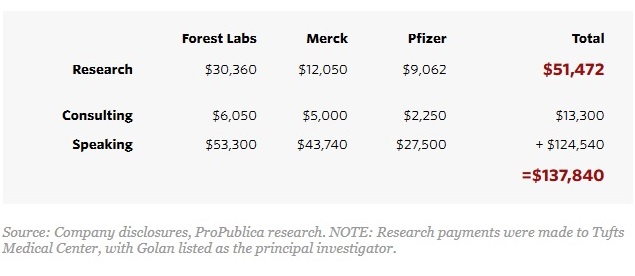
Golan, an infectious disease specialist, was the only doctor who received speaking, consulting, and research payments from three companies in 2012, the most recent year for which data has been compiled. Pfizer, Merck, and Forest Labs gave Tufts $51,000 for his research that year, in addition to paying him $125,000 to speak about their drugs and $13,000 for consulting. His speaking fees ranked second nationally among all the researchers examined, and his total personal payments ranked fourth.
Golan referred questions to the public relations department at Tufts Medical Center, which said in a statement that Golan complies with its research conflict-of-interest policy and that officials keep a close watch over his work.
“Dr. Golan’s work has contributed to the development of two important antibiotics, including the first antibiotic developed in the past 25 years to treat the growing threat of deadly C. difficile,” the statement said.
Pharmaceutical companies’ payments for promotional speaking and consulting appear to have decreased in recent years, as blockbuster drugs have lost patent protection and the push for transparency has advanced. Beginning this fall, all drug companies will have to publicly disclose payments they made to doctors, under the Physician Payment Sunshine Act, part of the 2010 Affordable Care Act.
But industry-backed clinical studies, which can lead to advances in care, have largely been seen as a separate matter.
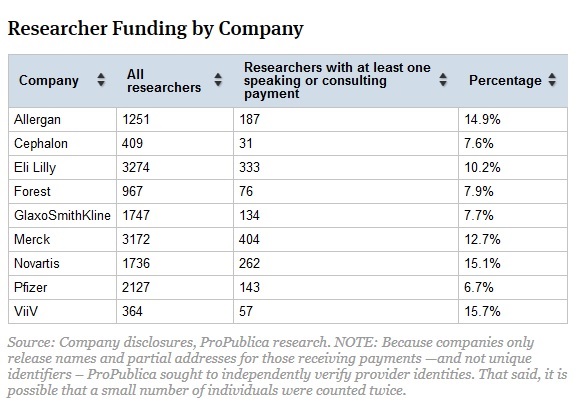
ProPublica’s is the first large-scale analysis of how frequently researchers receive additional payments from companies that fund their clinical trials. About 10 percent of researchers for the nine companies examined for this story also received money for speaking or consulting, or both.
Dr. Yoav Golan, an infectious disease specialist at Tufts Medical Center, received speaking, consulting and research payments from three companies in 2012, the only physician in ProPublica’s Dollars for Docs database that met those criteria. Some ethicists question doctors’ abilities to stay impartial when receiving both research and personal payments from pharmaceutical companies.
Pfizer had the lowest rate of dual relationships among its researchers, about 7 percent; Novartis and ViiV Healthcare had the highest, at more than 15 percent.
ViiV spokesman Marc Meachem said his company focuses exclusively on HIV medications, so “the number of people with the expertise to do both the research and be expert speakers is a lot smaller.”
In a statement, Novartis said it abides by the policies of different academic institutions, and requires doctors it works with to receive permission, if needed, from their employers.
The Mayo Clinic and University of California San Francisco prohibit employees from receiving personal compensation from companies that concurrently fund their research. Harvard allows doctors to take no more than $10,000 annually in personal income from companies funding their research.
“It’s such an enormous conflict of interest to have personal financial gain from the company that’s sponsoring a clinical trial on human subjects that you’re the principal investigator on,” said Lisa Bero, a professor of pharmacy at UC San Francisco who chaired its conflict of interest committee from 1999 to 2010.
Tufts University School of Medicine does not bar overlapping payments, but has a policy banning faculty members from speaking for a pharmaceutical company if “the company controls the content of the presentation, which may include creating or having final approval over the slides or presentation materials or setting limits on the scope of discussion.”
The companies that employ Golan as a speaker say they do create and have final approval of any materials used, but because he is an unpaid “volunteer faculty member” at the medical school by virtue of his hospital job, its rules don’t apply to him, a spokeswoman said. Tufts Medical Center requires only that doctors agree that the slides provided by companies for their lectures are scientifically accurate; Golan’s slides are reviewed by his department chairman as well.
Beyond his work for Pfizer, Merck, and Forest, Golan has reported receiving research, consulting, and speaking payments from Cubist Pharmaceuticals of Lexington. He co-wrote a 2011 research study, published in the New England Journal of Medicine, about the antibiotic Dificid with employees of its maker, Optimer Pharmaceuticals — a company purchased by Cubist last fall.
According to the most recent disclosure statement Golan submitted to Tufts Medical Center, covering the 12 months before March 4, 2014, Golan said he received a total of $260,000 to $359,995 in salary, speaking, consulting (and some travel) fees from Forest, Merck, Pfizer, Optimer, and Cubist. That does not include grants for research and is on top of what he earns for treating patients, according to Tufts which provided the data to ProPublica.
Tufts Medical Center said it has a committee with the authority to bar doctors from accepting additional payments from companies that fund their research if panel members conclude there’s no other way to mitigate the conflict of interest.
Instead, hospital officials have put Golan under a “rigorous management plan to ensure the research is done in a transparent and ethical manner, including meetings between Dr. Golan and a committee of research and physician leaders every six months to review his research and any changes in his relationship with industry,” the center said in a statement.
Ethicists and experts on conflicts of interest say overlapping payments can give researchers an incentive to bend or break the rules, sometimes in ways they aren’t even aware of. They note that published research supported by pharmaceutical companies tends to be more favorable to the drugs being studied than research funded by other sources.
Some questioned whether it was possible to balance a commitment to research with outside work for pharmaceutical companies.
“I would argue that any academic that has the time to be a part-time drug salesman needs to have a talk with their department chair right away about how they’re spending their time,” said Eric Campbell, a professor of medicine at Harvard Medical School who studies the topic. “If doctors want to be drug salespersons, they should go to be drug salespersons.”
Not every doctor who accepts research and promotional money is an academic. Some work for hospitals not tied to universities; others run their own clinical trial centers, and others are solo practitioners.
Dr. Andrew Wachtel is co-director of a research group called the Southern California Institute for Respiratory Diseases. He practices at Cedars-Sinai Medical Center in Los Angeles, but said he is not employed by the hospital. While conducting research for Merck, GlaxoSmithKline and Forest Labs, he received nearly $110,000 in 2012 for speaking on behalf of their products (the vast majority was from Forest).
“If I was doing research and speaking exclusively for one company, that might be construed as a conflict, and it is potentially a conflict,” he said. Working with multiple companies, he added, shows “I don’t have a personal gain by promoting one over the other.”
Still, Wachtel said that if he were given the choice between research and speaking, he would choose research. “I would just stop lecturing. It’s not a huge part of what I do or that big a deal.”
Dual relationships have come under scrutiny from lawmakers and regulators in the past. In 2009, Sen. Charles Grassley (R-Iowa) asked the National Institutes of Health to investigate university physicians who were paid by Merck to work on a campaign for the cholesterol drug Vytorin even though an internal study had showed it was no more effective than cheaper drugs. The relationships were first reported by the Chronicle of Higher Education.
Grassley raised particular concerns about Baylor College of Medicine, which didn’t tell the NIH that one of its researchers, Dr. Christie M. Ballantyne, had accepted $34,000 from Merck to consult about Vytorin while also receiving money from the NIH to study cholesterol-lowering therapies.
NIH director Francis Collins told Grassley in a letter that while Ballantyne had followed the rules and notified Baylor, the institution had not shared the information with NIH as it should have.
Even after Baylor said it tightened its practices, Ballantyne’s work with Merck continued. In 2012, ProPublica data shows, Ballantyne received speaking and consulting money from Merck while conducting research on the company’s behalf. Merck data shows he received $11,000 as a speaker and $7,175 as a consultant in 2013 as well.
Ballantyne said in an email that he is no longer participating in speakers’ programs because companies insist on having the final say on the slides used. “The position of the companies is clearly not consistent with the position taken by our medical school, and most other medical schools, so academic physicians almost never participate in speakers bureaus,” he wrote.
Baylor spokeswoman Lori Williams said Ballantyne spoke internationally on behalf of Merck in 2012 and 2013 and used his own slides, which did not violate Baylor’s rules.
Ballantyne wrote that he does not see a conflict in continuing to work as a consultant for companies that support his research. “I consult for companies because I have expertise that I hope may help in the search for new therapies. Research saves lives. I participated in a clinical trial when I was treated with lymphoma. If I had been diagnosed 50 years ago I would have lived less than 6 months.”
Ballantyne was one of several Baylor faculty members who received speaking or consulting payments in 2012 from companies that fund their research.
Baylor’s senior vice president of research, Dr. Adam Kuspa, said the school stepped up its review of research conflicts after the NIH findings. He said faculty are not permitted to serve as company speakers if a drug maker retains final approval of slides, but can spend up to 20 percent of their time on consulting.
Kuspa cautioned against going too far to regulate payments to doctors. “You certainly wouldn’t want people who didn’t know anything about the drugs consulting about what Pfizer’s doing in that area,” he said.
The Pharmaceutical Research and Manufacturers of America, the industry trade group, said it sees benefits in doctors working with companies in multiple roles. A company could hire a researcher overseeing a clinical trial, for example, to consult with other trial sites about patient recruitment or to speak about new uses for drugs being studied, officials said.
Kendra Martello, PhRMA’s deputy vice president of strategic operations, said research integrity is protected by institutional review boards, which are appointed by hospitals to approve the design of studies and patient consent forms, as well as by the U.S. Food and Drug Administration, as part of the drug approval process.
Massachusetts has taken a harder line than most states on doctors’ conflicts of interest. For a time, it banned meals to physicians. The state requires all pharmaceutical companies to disclose payments to Massachusetts doctors, though not for research, and posts the information online.
Policies at academic medical centers in Massachusetts differ. While Harvard caps the outside income of researchers, UMass Memorial Medical Center, Tufts Medical Center, and Boston University do not. They require researchers to disclose dual relationships internally but do not limit them as a rule.
Even so, John Randolph, vice president and chief compliance officer of UMass Memorial Health Care, said his conflict-of-interest committee rarely allows researchers much leeway.
“We are very conservative about it,” he said. “I don’t think we’ve allowed any faculty member to have a relationship like this where we’ve approved greater than $5,000 of activity from the same company — and that was only in the situation where the research and the consulting were in completely different areas of the company.”
ProPublica news applications fellow Eric Sagara contributed to this report.